So how are scientists going about developing the COVID-19 vaccine?
In our previous article we took a look at why a vaccine for COVID-19 is crucial and when one will be available to the general public.
We also mentioned that many innovations are being tried out to speed up the development of new vaccines.
Here, let’s take a deeper look at the various approaches and technologies being used in developing the COVID-19 vaccine.
This article will cover:
- Vaccine development approaches – traditional vs new approaches.
- Supplementary vaccine development technologies.
- Why multiple approaches must all be employed for developing the COVID-19 vaccine.
The History of Inoculation and the First Vaccine
In 1796 English physician Edward Jenner inoculated a young boy against smallpox with a method that is widely considered the first vaccine.
But the first written accounts of humans using artificial means to induce immunity, i.e. inoculation, come from China.
Chinese texts from the 17th and 18th centuries refer to relatively advanced forms of smallpox inoculation, and some texts from even earlier refer to more rudimentary methods.
Since Dr. Jenner’s first vaccine, several different approaches to making a vaccine have been developed.
Let’s take a look at several older and more recent approaches being employed to develop the COVID-19 vaccine.
Vaccination
The purpose of a prophylactic vaccine is to train our immune system to be able to recognize a pathogen and quickly launch the required immune response to neutralize it.
Traditional vaccines against viral diseases consist of a weakened or inactivated form of the whole virus, or parts of that virus. In recent years genetic engineering is being used to create new types of vaccines which include synthetic materials developed using the genetic sequence of the virus.
Once a vaccine is introduced into our body the immune system will respond to it in a way similar to how it would respond to the corresponding wild virus; it will begin to produce tailored antibodies to neutralize it.
Unlike the wild virus, however, the vaccine does not cause harmful infections. So our immune system has more time to respond effectively.
After antibodies neutralize the invader our immune system will create and store Memory B cells that can identify the wild virus in the future. The ability to ‘remember’ the virus is called immunologic memory.
If we are later exposed to the wild virus our immune system will quickly recognize it and produce sufficient quantities of the required antibody. This head-start generated by the vaccine allows our body to neutralize the wild virus before we contract a disease such as COVID-19 in the first place.
Immunity developed with a vaccine can last anywhere from a few months to a lifetime.
Humoral and Cell-mediated Immunity
The ability to produce antibodies to prevent an infection is called humoral immunity. Humoral refers to our body fluids which the virus first invades and where antibodies circulate.
Some vaccines can also produce cell-mediated immunity (CMI). After a virus circulating in our body fluids successfully invades a cell the cell will respond by creating T cells which can produce inflammatory and antiviral agents to destroy the invader inside of it.
Similar to humoral immunity, as the invader is cleared Memory T cells will be created, ready to support T cell production once the wild virus enters cells in the future.
Traditional Vaccines
Types of traditional vaccines include:
Live Attenuated Vaccine
This vaccine uses a live, but weakened form of the virus. Weakening the virus can be accomplished through repeated culturing.
The attenuated virus can infect cells and replicate inside the body. The infection, however, is much less severe than that caused by the wild virus.
The immune response closely mimics a natural response to a wild virus. So often only one dose of the vaccine is enough.
Both humoral and cell-mediated responses could be produced. So the immunity it builds can last for many years or even a lifetime.
As it is still a live virus, however, it could cause side effects for people with compromised immune systems. In extremely rare cases the attenuated virus could also revert to its dangerous state, leading to harmful infections. This is known as ‘risk of reversion’.
Therefore, extensive research and testing, often over many years, is required before a live attenuated vaccine can be declared safe and deployed to the general public.
Inactivated Vaccine
An inactivated vaccine contains a dead version of the virus, and therefore is also called a ‘nonlive vaccine’. The virus can be killed in various ways such as with heat or chemicals. Such vaccines are safer than live attenuated vaccines, but less effective.
The inactivated virus cannot replicate inside our bodies, so multiple doses are often required.
Inactivated vaccines mostly cause humoral immunity and seldom cause cellular immunity.
Developers of Inactivated Vaccines for COVID-19
The following companies and research institutes are developing Inactivated vaccines for COVID-19:
Subunit Vaccines
Scientists realized that the immune system was actually responding to a part or parts of the virus; mainly proteins on its surface. The immune system creates antibodies that correspond to the shape of these proteins so as to latch onto them before they can bind with our cells.
So vaccines were developed consisting of only such proteins, known as antigens, instead of the complete virus. These vaccines are called Subunit vaccines.
For SARS-CoV-2, the virus which causes COVID-19, it is the spike protein on its surface that it uses to break into human cells which many scientists consider as the key part to be targeted. So this Spike protein has been chosen as the antigen to be used for developing many COVID-19 vaccine candidates.
A Subunit vaccine can be classified as a type of Inactivated vaccine as it also can not replicate inside the body. Like other Inactivated vaccines, therefore, Subunit vaccines are inherently safe.
However, eliciting the desired immune response is not so easy for Subunit vaccines. When proteins are introduced by themselves they are not stable. They could easily change shape and, therefore, the antibodies elicited might not be so effective.
Even if useful antibodies are produced, the immune response is not as strong as that produced by live vaccines so multiple doses could be required to maintain immunity.
Viral-Like Particle Based Vaccines (VLP)
Viral-like particle (VLP) vaccines are a type of Subunit vaccine. Instead of choosing just one or more proteins from the virus, however, VLPs mimic the structure of the virus’s protein capsid (shell). Multiple proteins are presented in the same structural shape as they are found on the wild virus, but without the genetic code on the inside of the virus.
As Dawn Bowdish, Canada Research Chair in aging and immunity at McMaster University in Canada explains “A virus-like particle looks like the outside of a virus but doesn’t have any of the genetic material on the inside. It doesn’t cause infection but looks really similar from your immune system’s perspective.”
As VLP vaccines elicit a more natural immune response they can be more effective in building cell-mediated immunity than other Subunit vaccines.
VLP technology has created several successful vaccines since the 1980s including those against hepatitis B, cervical cancer and hepatitis E.
Developers of VLP Vaccines for COVID-19
The following companies and research institutes are developing VLP vaccines for COVID-19:
- Medicago (Canada)
Weaknesses of Traditional Vaccines
These traditional vaccines have helped humanity to eliminate or control many deadly diseases over the years.
However, these approaches are time consuming and costly due to:
- Vaccine developers need to work with the live pathogen, which requires many safety measures to be taken.
- The development and manufacturing of each vaccine must take place with dedicated equipment and possibly inside of dedicated facilities to prevent contamination from other vaccines.
- Vaccine manufacturers need to grow enough of the pathogen to manufacture the required number of doses of the vaccine. Often the virus or antigen is grown in eggs which can take up to 6 months.
- Many of these vaccines require refrigeration so are difficult to store and transport.
New Approaches for Developing the COVID-19 Vaccine
Several new approaches and technologies have been developed in recent years that solve many of the drawbacks associated with traditional vaccines.
All of these innovations are being tried out to speed up the development of numerous vaccine candidates for COVID-19.
Vaccine Platforms
In order to speed up development of vaccines to fight new pathogens the research community has been developing vaccine platforms. A platform has the basic components for creating a vaccine and can be easily adapted to create new vaccine candidates. Scientists, therefore, do not need to start from scratch each time a new vaccine begins development.
Platforms that were used to develop vaccine candidates for other coronavirus diseases including SARS and MERS in recent years are now being adapted for developing the COVID-19 vaccine.
These platforms are also referred to as:
- Rapid-response platforms – referring to the fact that the platform is quickly adaptable to be able to rapidly develop various new vaccines.
- Plug and Play vaccine design – referring to how different antigens or genetic material can be ‘plugged’ into the same platform to start ‘playing’ with new vaccine candidates.
Viral Vector Vaccines
Viral vector vaccines use genetic engineering to create hybrid viruses. Genetic code from a dangerous virus is inserted into the genome of a harmless virus to create the vaccine. The harmless virus is known as the ‘vector’, ie. deliverer.
When the vaccine is injected into the recipient it expresses properties of the dangerous virus which is recognized by the immune system and results in the desired immune response.
For COVID-19 the genetic code of the spike protein on the surface of the SARS-CoV-2 virus is often chosen to add to the vector virus.
There are several terms related to this viral vector technology including:
- Chimeric virus: The hybrid virus created by combining genetic information from a dangerous virus with the harmless vector.
- Recombinant – referring to the process of creating this chimeric virus by recombination; i.e. exchanging pieces of genetic material from one virus into another.
Advantages of Viral Vector Platforms
- There is no need to obtain a live virus to start the vaccine development work.
- The vector virus and the synthetic viral proteins from the dangerous virus are both harmless.
- The chimeric virus can be delivered in live form without the need to apply chemical inactivation. Therefore, a more natural immune response can be induced.
- The chimeric virus can replicate inside the body. Cell-mediated immunity (CMI), therefore, could also be elicited.
- The replicating gene of the vector virus could be deleted to make the vaccine even safer.
- The chimeric virus can be grown quickly in cell cultures.
- Common production facilities can be used to manufacture multiple vaccines.
Disadvantages of Viral Vector Platforms
- If the recipient has pre-existing immunity to the vector virus it could be destroyed before it can induce an effective immune response.
Developers of Viral Vector Vaccines for COVID-19
The following companies and research institutes are developing Viral Vector vaccines for COVID-19:
- University of Oxford (UK)
- CanSino Biologics (China)
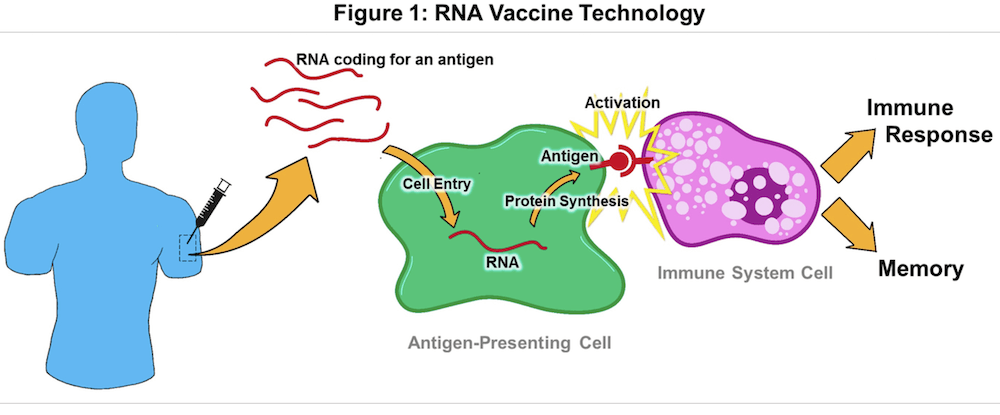
Nucleic Acid Vaccines
Rather than deliver an antigen into the body, Nucleic Acid Vaccines directly inject DNA or RNA code from the virus into our cells. Our cells then follow the code to produce the proteins; i.e. the antigen, themselves. Our immune system then reacts to the antigen just as if it was part of the real virus.
With DNA vaccines, after the injection the cell’s nucleus first transcribes the DNA to produce messenger RNA (mRNA) molecules which carry instructions to the cell’s cytoplasm to make the specific protein(s).
RNA Vaccines go one step further by injecting the mRNA molecules directly. A RNA vaccine could be safer than a DNA vaccine as it does not interfere with the DNA of the host cell.
Advantages of DNA and RNA Vaccines
- Fully synthetic manufacturing process – no need to obtain a live virus to start vaccine development or use any live virus in the manufacturing process.
- The same facilities can be used to develop and manufacture multiple vaccines.
- As the antigen is created inside a cell, both humoral and cell-mediated immunity could be elicited.
- Storing nucleic acid vaccines is easier than traditional vaccines.
Weaknesses of DNA and RNA Vaccines
- In a BBC interview, Robin Shattock from the Department of Infectious Diseases at Imperial College London, one group working on a RNA vaccine for COVID-19, suggested that using a more complex approach for developing the COVID-19 vaccine (presumably the live-attenuated or Inactivated approaches mentioned above) could lead to longer term immunity than this new approach.
- There is as yet no nucleic acid vaccines licensed to be used on humans, so it is an unproven technology.
- Delivery of the RNA molecules directly into cells can be a difficult process as RNA easily degrades.
Developers of Nucleic Acid Vaccines for COVID-19
The following companies and research institutes are developing Nucleic Acid Vaccines for COVID-19:
DNA Vaccines:
- Inovio (US)
mRNA Vaccines:
- Moderna (US)
- BioNTech (Germany)
- CureVac (Germany)
- Imperial College of London (UK)
Supplementary Technologies for Developing the COVID-19 Vaccine
Some universities, research institutes and companies have developed supplementary technologies to deal with the innate weaknesses of these various approaches. These new technologies are being used to develop several COVID-19 vaccine candidates.
Molecular Clamp Technology
As mentioned above, when developing Subunit vaccines it can be difficult to ensure the proteins of the virus remain stable when expressed on their own.
The University of Queensland in Australia (UoQ) has developed a proprietary technology called a Molecular Clamp to help solve this issue. This process synthesizes the chosen protein(s) and then clamps them into shape so when presented to the immune system it creates effective antibodies.
The RNA Printer™ Manufacturing Facility
CureVac, a biotechnology company from Germany, is developing a Nucleic Acid Vaccine for COVID-19 with its mRNA platform technology. This program, like the one from UoQ, was one of the earliest to receive financial support from Coalition for Epidemic Preparedness (CEPI) alliance for developing a vaccine for COVID-19.
CureVac has several hundred patents related to overcoming the instability of mRNA molecules and manufacturing mRNA vaccines.
CureVac has patented a unique down-scaled, automated messenger RNA (mRNA) printing facility. The RNA Printer™ can be easily transported to areas of need and manufacture more than a hundred thousand doses of an mRNA vaccine within just a few weeks.
Core Shell Structure Platform
A Chinese company, Shanghai-based Stemirna Therapeutics, has patented a core shell structure platform (LPP/mRNA®) to protect the RNA molecules as they are delivered into the body.
Trimer-Tag Technology
Another Chinese company, Clover Biopharmaceuticals of Chengdu, has developed a patented technology called Trimer-Tag which is able to identify which antigens develop the most effective antibodies. As viruses have a number of surface proteins, this will help scientists to choose the genetic code of the protein which produces the most effective antibody.
Clover has already used this technology to detect antigen-specific antibody when tested on the blood of recovered COVID-19 patients.
Plant-Based Platform
A Canadian company, Medicago, is using its plant-based platform rather than chicken embryos to grow virus-like particles for its COVID-19 vaccine. This will vastly speed up the antigen production process.
Vaccine Adjuvants
Capacity for manufacturing the COVID-19 vaccine will be a big issue. If the virus is still spreading when a vaccine is approved hundreds of millions of doses could be required to protect the entire world’s population.
One way to increase the number of doses of vaccine available is to make use of what is called an adjuvant.
GSK, a British multinational pharmaceutical company, has signed an agreement to allow all CEPI supported programs to make use of its adjuvant technology in developing the COVID-19 vaccine.
As GSK’s website explains “An adjuvant is added to some vaccines to enhance the immune response, thereby creating a stronger and longer lasting immunity against infections than the vaccine alone. The use of an adjuvant is of particular importance in a pandemic situation since it can reduce the amount of antigen required per dose, allowing more vaccine doses to be produced and made available to more people.”
GSK is also providing its adjuvant technology to Clover Biopharmaceuticals of China for use in their COVID-19 vaccine.
Multiple Approaches Must All Be Employed
Not all of the recent innovations in vaccine development have been used to create successful vaccines. We can not, therefore, rely on just one approach even if it would, theoretically, win the race to develop a COVID-19 vaccine.
Moreover, even if one approach is successful in developing an effective vaccine before others, it does not mean that continuing to develop vaccine candidates with other approaches should be stopped.
The earliest available vaccine might produce immunity that lasts only a short period of time, whereas other approaches might result in longer term immunity.
A vaccine which can induce both humoral and cell-mediated immunity is usually preferred. Cell-mediated immunity, however, seems to be less important for combating some viruses, and we are not yet sure if it is critical for a COVID-19 vaccine.
Factors including, age, gender, race, ethnicity as well as vaccine dosage all affect how different people respond to different types of vaccines.
Dr. John Shiver, Sanofi Pasteur’s senior vice president of global vaccine research and development stated on March 2nd, 2020: “It’s great that new and multiple technologies are being tried” for developing the COVID-19 vaccine, and “The more approaches being tried will increase our chances of success.”
Fortunately, there are already over a hundred COVID-19 vaccine development programs using various approaches and technologies underway around the world. Several of these programs already have candidates in human trials as the race for a COVID-19 vaccine heats up!
Chinese Health officials are supporting multiple programs using different approaches just within this one country. Zeng Yixin, deputy director of the National Health Commission, said their scientists are currently testing five kinds of vaccines; live attenuated vaccine, inactivated vaccine, live vector vaccine, recombinant vaccine, and nucleic acid-based vaccine.
A type of COVID-19 vaccine could be preferred for different reasons. For instance, one that is easy to administer might be preferred for children even if it only provides short-term immunity. Another could be required for seniors if it is the only one they effectively respond to. And some types could be more suitable for some countries if they are easier to transport and store.
In the end, we could end up with a number of different COVID-19 vaccines made available around the world. Considering that billions of doses of the COVID-19 vaccine need to be manufactured in the next few years, this would be a good thing!